Aims: It were compared that changes in the cost of drug clinical trials on both amount and structures between 3 years before and after self-examination and inspection initiated by the CFDA at July 22, 2015 in China.
Methods: The paper Identify the increase and composition in cost change, analyze the impact of the cost of
new CRC, the labor service of researchers, the audit company, the institutional drug management and quality control on the quality improvement of drug clinical trials.
Results and Conclusions: According to the article, the emergence and rise in most clinical trials costs are conducive to the quality enhancement of drug clinical trial, the occurrence and continuous increase of CRC costs improve the quality, at the same time, It implies a barriers factor to improve the drug clinical trial quality in China recently years.
Inspection of Drug Clinical Trial, cost, Quality, SMO (Site Management Organization), CRC (Clinical Research Coordinator)
On July 22, 2015, China Food and Drug Administration issued Announcement of Self-examination and Inspection of Drug Clinical Trial Data . As a thunder, it opens what the industry calls the 7.22 storm. With "the most rigorous standards, the most strict supervision, the most severe punishment, the most serious accountability", the most stringent drug registration self-examination and inspection in history was launched. Great changes have taken place in the field of drug clinical trials With the deepening of self- examination and inspection in China in the past three years, among which the cost of clinical trials is one of the important changes.
The purpose of this paper is to analysis the quantity and structure change of clinical trials cost before and after 7.22, explore the corresponding relation between cost change and quality improvement.Both cost and quality changes need multiple cause to one effect ,and the two changes as initiator appeared one cause to multiple effect.
When two kinds of changes interact with each other alternately, there is a positive correlation interdependent between the changes, In the meantime, which also implies the hidden worry of deviating from the original intention at the increase of investment corresponding the improvement of quality.
There are different visit procedures and difficulty level in different drug clinical trial protocols. In recent years, diabetes, cancer drugs were frequently carried out in clinical trials field, because the input costs of Similar drugs are comparable, Take them for example, we can analyse the change of the amount and composition of the clinical trials cost. In this paper, data come from The First Affiliated Hospital of Wenzhou Medical University, Tianjin Medical University General Hospital , Xiehe Hospital Tongji Medical College Huazhong University of Science and Technology. Since multiple-center clinical trials of drugs are generally signed contracts at the same price at every centers, the three hospitals can represent the average cost level at the same period in China.
This paper focuses on the costs involved in the agreements signed by the hospital during innovative drug phase II, III and generic drugs random controlled clinical trials, make some analysis on the monitoring, audit and tax input of the sponsor. It does not involve the costs of phase I clinical trials, clinical trials data management statistics, cold chain logistics, and researcher meetings. The unit of measurement is RMB (RMB yuan).
Change trend of clinical trial cost from 2013 to 2018 (taking diabetes glycemia controlling drug as an example)
The paper select the program which have similar visiting frequency and observation period, compare the costs of the of clinical trial agreement signed three years before and after July 22, 2015. (note: in the chart below, the cost of 2015 is before July 22, after7.22 the overwhelming majority of Chinese hospitals had endured a shock period for about half a year in contract signing)
The change of global clinical trial cost during 2013-2018: During 2013-2018, each cases innovative glycemia controlling drugs cost amounts and composition of United States and European countries signed by hospital clinical trial agreement in the the territory of China are shown, as (Figure 1).
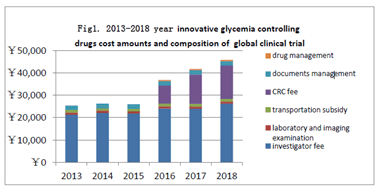
Figure 1. 2013-2018year innovative glycemia controlling drugs cost amounts and composition of global clinical trial
Figure 1: The most significant change of absolute value and the proportion is paid to the SMO's (Site Management Organization) CRC (Clinical Research Coordinator). 2013-2018, the average amount respectively: 0, 0, 0, 8052, 13154, 14865,it presented remarkable change before and after7.22, and have a increasing trend year by year. The average amount of the investigator fee from 2013 to 2018 was 21312, 22086, 21830, 24055, 23898 and 26219 respectively, which general change is not obvious and the slight increase is related to inflation. The average amount of subjects research relevant laboratory and imaging examination respectively in 2013-2018: 935, 968, 985, 992, 987, 997, that barely changed because the price of examination fee is set and changed by hospital are approved by the price authority, The average amount of subjects transportation subsidy from 2013 to 2018 respectively is: 1150, 1097, 1116, 1235, 1184 and 1183,the transportation subsidy usually is between 150 and 200 for a single visit. The documents management fee remains unchanged for 6 years at 2000 per case for 15 years. The drug management fee has been charged since July 22, 2015 at 500 per case, which obvious but small change on amounts .
The change of Chinese clinical trial cost during 2013-2018: During 2013-2018, each cases generic glycemia controlling drugs cost amounts and composition signed by hospital clinical trial agreement in the the territory of China are shown, as (Figure 2).
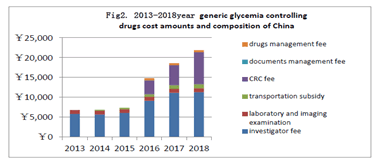
Figure 2. 2013-2018year generic glycemia controlling drugs cost amounts and composition of China
The most significant change of absolute value and the proportion is paid to the CRC of SMO company. 2013-2018, the average amount respectively: 0, 0, 0, 3530,5065,8120,it presented remarkable change before and after7.22, and have a increasing trend year by year. The average amount of the investigator fee from 2013 to 2018 was 5800,5675,6035,9126,11112, and 11293 respectively ,which general change is obvious . The average amount of subjects research relevant laboratory and imaging examination respectively in 2013-2018: 965,983,968,972,969,991,that barely changed because the price of examination fee is set and changed by hospital are approved by the price authority. The average amount of subjects transportation subsidy from 2013 to 2018 respectively is: 0,200,330,650,950,960,cost of single visit wasn’t counted and a nominal fee was paid when the clinical trial was completed , or there was n‘t transportation subsidy before 7.22.it was ranging from 100 to 150 after 7.22. fee of documents management wasn’t counted. The drug management fee was 500 per case beginning after July 22, 2015.
Comparing figure 1 and figure 2, The investigator fee and cost of CRC in global multiple-center clinical trial is significantly higher than that fees of China . Besides the gap of sponsor economic power, the global multiple center clinical trials focus on innovative drug while the majority of China clinical trials are generic drugs clinical trials.
Composition and proportion of clinical trial cost amounts 3years before and after 7.22 (taking lung cancer drugs as an example)
Cancer drugs are common varieties of drug clinical trials in recent years, so the cost of cancer drugs clinical trials are representative. we take the cost of lung cancer drugs which there are 18 to 20 visits excluding the lifetime visit and unscheduled visit as example, compare the cost of clinical trials before and after July 22, 2015.
Composition and proportion of global clinical trial cost:
- Composition and proportion of global clinical trial cost 3 years before July 2015 within China: The average of 2013-2015 was showed as Fig 3. Investigator fee 30697, 64.09% of the proportion, the subjects examination fee 11847, 24.73% of the proportion, the subjects traffic subsidy 3355, accounting for 7.00%, documents management fee 2000, accounted for 4.18% (Figure 3).
- Composition and proportion of global clinical trial cost 3 years after July 2015 within China
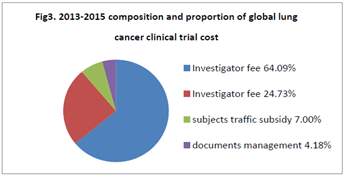
Figure 3. 2013-2015 composition and proportion of global lung cancer clinical trial cost
The average of 2016-2018 was listed as Fig 4. Investigator fee 40187, 44.37% of the proportion, the subjects examination fee 12053,13.31% of the proportion, the subjects traffic subsidy 4200, accounting for 4.64%, documents management fee 2000, accounted for 2.21%, drug management fee 500, accounting for 0.55%, CRC fee 31624, accounted for 34.92%. (Figure 4).
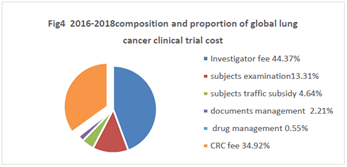
Figure 4. 2016-2018composition and proportion of global lung cancer clinical trial cost
Composition and proportion of Chinese clinical trial cost:
- Composition and proportion of Chinese clinical trial cost 3 years before July 2015 within China: As Fig 5,the average of 2013-2015 was displayed. Investigator fee 18930, 71.91% of the proportion, the subjects examination fee 5395, 20.49% of the proportion, the subjects traffic subsidy 2000, accounting for 7.60%, documents management fee not is charged (Figure 5).
- Composition and proportion of Chinese clinical trial cost 3 years after July 2015 within China
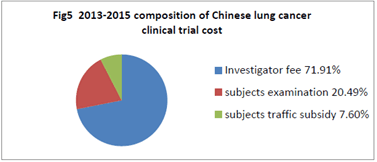
Figure 5. 2013-2015 composition of Chinese lung cancer clinical trial cost
The average of 2016-2018 was showed in Fig 6. Investigator fee 23400, 47.85% of the proportion, the subjects examination fee 6252, 12.78% of the proportion, the subjects traffic subsidy 2250, accounting for 4.60%, drugs management fee 500, accounted for 1.00%, CRC fee 16500, accounted for 33.74%, documents management fee not is charged (Figure 6).

Figure 6. composition of Chinese lung cancer clinical trial cost
No matter comparing figure 1 and figure 3, figure 4 of global clinical trial or comparing figure 2 and figure 5, figure 6 of Chinese clinical trial,the composition and proportion cost are similar between the glycemia controlling and cancer drug clinical trial.One difference is that the cancer drugs clinical trial involve more imaging and laboratory examination, so which have higher costs and a larger proportion on fees.
Analysis above cost change
Investigator fee: The increase of investigator fee of global clinical trial cost is not obvious, eliminating inflation factor, the amount almost a little change from fig1.Fig2 shows the cost of Chinese multiple-center site grew obviously and It's up 30-40% after 7.22.
CRC fee: CRC fee Is the most significant part of the change., There were few CRC fee expenditures before July 22 2015,but it increased starting from 2016 to 2018 year by year. As fig1, the amount and proportion of CRC fee of global multiple-center glycemia controlling drugs 2016-2018 are respectively 8052 (22%), 13154 (32%), 14865 (32%). As fig2, the amount and proportion of CRC fee of Chinese multiple-center project of glycemia controlling drugs 2016-2018 are 3530 (24%), 5065 (27%), 8120 ((37%).Fig 4 shows the mean of global multiple-center cancer drugs clinical trial CRC fee from 2016 to 2018 was 31624 and proportion was 34.92%.Fig 6 shows the mean of Chinese multiple-center cancer drugs clinical trial CRC fee from 2016 to 2018 was 16500 and proportion was 33.74%.
Drugs management fee: The 7.22 incident promoted the standardized management of drugs in GCP central pharmacy and the process of drug clinical trials, due to the low amount, only about 1% of the total investment in drug clinical trials.
Documents management fee: There are some differences different hospitals according to Chinese and global clinical trials. In terms of Good Clinical Practice of Pharmaceutical Products [SFDA order No. 3, Effective on September 1, 2003] Article 52:” The investigator shall keep the documents of the clinical trial for five years after the completion of the trial” [1]. However, when the global drugs clinical trial finished, the documents are generally required to keep for 15 years, the documents global trial was preserved freely with reference to China’s domestic the first 5 years when it was completed, the charge standard of 6 to 15 years, on the basis of the done cases amount and documents size, hospitals charge 1500-5000 every year. Toward the documents after 5 years finished Chinese clinical trial if the it were handed over to a qualified third party specified by sponsor, although hospitals will continue to be stored for inspect, rarely charge fee.
Related cost changes not covered by the clinical trial agreement before and after the 7.22 incident
Change is obvious in monitoring and audit mainly on the inputs of human
activity. After 7.22, along with CFDI inspect strongly in concomitant drugs Adverse Events (AE) and serious adverse events(SAE) of subjects, monitor need to check the Hospital Information System(HIS), Laboratory Information System(LIS), Picture Archiving And Communication System(s PACS)of hospital in the process of monitoring, to ensure that a combination drug, especially a combination drug prohibited by the protocol, AE and SAE are recorded Completely without omission. In recent years, the risk-based monitoring advocated by the sponsor has improved the monitoring efficiency certain to some degree, but in general, after 7.22, monitoring will spend 15-20% more time in the hospital, and the corresponding labor cost will increase 15-20%.
After 7.22, the attention paid by sponsor to the audit has been significantly increased, and the frequency of audit received by hospitals has increased significantly. Generally, sponsor pays the fees and entrusts a third party audit company to carry out the audit. Another change was tax. Because The State Administration of Taxation changed the Value Added Tax( VAT) rules two years ago in China and clinical trial cost in invoice billing system belonged to the scope of tax, when sponsors remit money to hospitals, the added 6% tax must be paid, there was a 6 percent increase in the total amount of money that the sponsor had to pay. The tax cost has nothing to do with the 7.22 event, but the time of the reformed tax law universal implementation was almost in line with the 7.22.
After the 7.22, both the global and Chinese domestic multiple-center clinical trials in China have changed, the change on clinical trials initiated by the Chinese domestic sponsors are greater than global clinical trials.
New phenomenon in industry recent three years: emergence of SMO and audit company increase of clinical trial price
SMO sprang up like mushrooms: SMO provides the study coordinator service, sends CRC to the hospital to assist in the implementation of clinical trial who performs the non-medical judgment task duties authorized by Principal Investigator (PI). SMO began to develop at 2008 in China, which had been always small scale until July 22 2015, the number of SMO expanded rapidly and staff of SMO surged, one grew from its establishment to a company with nearly 600 CRC within one year.
Centralized management the trial drug at center pharmacy: Professional technique department of the hospital is difficult to achieve dedicated clinical trial drugs management due to in sufficient experience on drug management and limited ability of facilities and equipment in the department not meet the inspection requirements when they undertake clinical trials. GCP center pharmacy equip centralized management of full-time pharmacists who passes through GCP training, which can be better to perform the standard operating procedures of trial drug receiving, storage, distribution, use, residual drug recycling and destruction, etc.
Emergence of audit company: After 7.22, third-party audit institutions sprung up successively which provide clinical trial training , consulting services, and audit comprehensively on-site of clinical trial data and records. Its independent examine mode makes the division of labor refined and specialized.
Cost growth boosting by Industry change
CRC fee growth: CRC fee growth was the most rapid and significant change in the cost of clinical trials which was clearly showed in figure 1-6.
Investigator fee: The increase of investigator fee are divided into explicit growth which can be clearly shown in figure 1-6 and implicit growth. Implicit growth refers to the addition of CRC, that part non-medical judge work was done by Investigator before is done by CRC now., the Investigators ' labor fee have increased in disguised form because the decrease of Investigators’ workload.
The fee of audit company: The audit company usually sends 5 persons to a site for 2 to 3 days on-site audit, and the sponsor needs to pay the audit company fee of 80 to 100 thousand yuan. The number of site which are audited shall be determined by the evaluation of the sponsor and the request initiating from the clinical trial site and consultation with the sponsor.
Drug management fee, quality control fee: The amount of drug management fee is small, and the fee of each subject generally will charge 200-500 yuan according to the complexity level of the clinical trial, which has played a good role in regulating the management of drugs in the implementation process of drug clinical trials. Before 722, the drug clinical trial research institutions it charged quality control fees is less, part of the drug clinical trial research institution begin charging quality control fees after 722. Fees charged by agencies vary and the overall amount is small. The fees earmark for its specified purpose only so play a relatively important role in quality control [2].
There is a saying in the industry about the cost and quality of clinical trials: high costs do not surely mean high quality, but low costs certainly cannot make the high quality. Low cost is difficult to guarantee the quality of clinical trials is the consensus in the industry, any step is needed labor and material resources budget. Research institutions could perform a quality clinical trials for sponsor with limited budget. On the other hand, there are many factors influencing quality. High investment and high cost do not necessarily guarantee high quality.
The price of clinical trials showed a sharp increase nearly 3 years, the quality of clinical trials have obvious change too, but not one to one correspondence between them, both changes are one cause and many fruit, many cause and one result. Price increase is necessary for quality improvement, but not a sufficient condition. Price increase is not positively correlated with quality improvement, certain factors raise prices to improve the quality in some ways, at the same time obstruct the quality improvement in other ways.
Most of the cost increases in clinical trials are positively correlated with quality improvements, but the boom in SMO needs to be split in two. The CRC service fee paid by the sponsor to SMO accounts for about 30% of the total investment in clinical trials, which can not to be underestimated. It is undeniable that the intervention of CRC has provided convenience and some assistance for investigator to improve the quality of clinical trials. However, both the offside of CRC and the absence of investigator, the deficiency of CRC ability and the excessive dependence of investigator on CRC exist simultaneously.
Absence and offside associated with CRC
After 7.22, the research institutions which undertake drug clinical trials were asked CRC for the trail at the time of signing the agreement with sponsor or Contract Research Organization (CRO).The CRC agreement that is a separately triple agreement generally independent from the main clinical trials agreement, signing by
sponsor (or CRO), SMO, hospital(research institutions). Since Chinese public hospital in financial management is not easy to send remittance to businesses like SMO, It is stipulated in the triple agreement that the sponsor or CRO shall remit money to SMO, and SMO shall send CRC with relevant professional background and GCP training to the hospital to assist investigator' work. CRC need to sign confidentiality agreement of the work content when the principal investigator authorize and assign to CRC who is occupied in non-medical judgment work. The person responsible for the work content that done by CRC are principal investigator, sub-investigator or general investigator, if CRC is incompetence, hospital(research institutions)has the right to require SMO to replace competence CRC.
The lack of industry training and supervision is impeding the rapid development of the SMO industry. New SMO registered spring up,at the same time, The shortage of CRC talent has led to a poaching war between SMO companies. CRC frequent job-hopping,impetuous atmosphere spreading within the industry, that result in a profit-oriented SMO company short - term development model. It is an ordinary state that training process before the post can be shorten or even free when possible. Another problem is that the management of CRC by SMO company is regulated long-distance supervision. The working districts of CRC are in various hospitals, and the location of SMO company is the registration district. It also makes supervision difficult [3].
The major of CRC practitioners is mostly undergraduate or even community college graduates in pharmacy or nursing and they are not very excellent individuals In terms of the quality of personnel in the same period same major, in addition, most of them enter CRC industry immediately after graduation, with no working experience and insufficient professional knowledge accumulation. When SMO were limited trained, many incompetent CRCS were sent to hospital to perform CRC functions.
In terms of hospital, when a CRC was sent to site, the investigators' attitude toward the quality of CRC work is : excellent CRC is very lucky, usually CRC is very normal, poor CRC is better than none, so we dare not to expect a good CRC, one is better than none.
On the one hand, CRCs’ capabilities have not to be improved but who have plenty of time to be fully at the mercy of the investigators yet, on the other hand, investigators who have been too busy with clinical affairs to leave clinical trials behind. In such a state, CRC performed a lot of investigator’s duties and even the principal investigators’. The work of principal investigators do, just is a know what were signed by the authorized table,And then were the principal investigator and investigator in a hurry to sign a large number of table without ask why and what when the CRC refers to where to sign. Other common phenomenon, the investigators’ account and password to login fill in EDC (Electronic Date the Capture System), authorized by the sponsor, were frequently dabbed down to CRC and investigators ignored to verify the process and date after filled out by CRCs. Signatures with no asking the reason, process and content and the behavior that giving the EDC account password to CRCs, as the investigator, there maybe no completed monitoring of the clinical trials whole process,and hidden dangers in drug clinical trials exits.
Sponsor and clinical trial institution kidnapped by SMO
Hospital or clinical trial institution require sponsor to equip the CRC for clinical trial, sponsor remitted the funds and entrusted SMO, and SMO provide CRC services send CRC to clinical trial site. Long-distance supervision from SMO to CRC works badly, the quality of CRC work is rated by the clinical trial site and this kind of evaluation tends to lag. In that situation there are no timely effective feedback. Once CRC's incompetence be discovered, a irreversible effects on the quality of clinical trial and impact trial schedule already exits there. As the sponsor of the bid and the SMO company long-distance supervision cannot effectively control the quality of CRC. When hospital clinical trials office managers and investigators find CRC can not conform to the requirements of the quality, if a replacement of CRC is required, The hospital side must count the time and opportunity costs of CRC turnover. There are risks, for example, the replacement CRC may also be incompetent, takes time for CRC get familiar work after replacement, between interactions with CRCs may also occur mutual buck-passing due to the work handover when problems arise. The current situation of shortage in CRCs and the scarcity of high quality CRCs has led SMO to “kidnap” bidders at the service price and “kidnap” Hospital and investigators at the service quality.
Exploring new CRC models
Besides the SMO provide CRC, individual hospital, such as zhongshan Hospital Affiliated of Fudan University, Beijing 301 Hospital, Hunan Cancer Hospital and Cancer Hospital Affiliated of Sun Yat-sen University , established their own CRC team to help themselves take up clinical trial.Ina word, The purpose is to create a group of high level and stable CRC team who can assist investigators to accomplish high quality drug clinical trial from the CRC service consignment outsourcing to SMO model to hospital training, employing and supervising CRC themselves. The hospitals own CRCs account for a small proportion of the total number of CRCs exported by SMO now.Two CRC source models of SMO supplying and hospital providing themselves have their advantages and disadvantages. The CRC sent by SMO can cover clinical trial site in cities across the country, but the personnel sent by SMO to hospitals need to have a process of run-in and acceptance by hospitals. The CRC team built in hospital has many advantages in local work, but it cannot support the clinical trial needs of other hospitals.
There are no universally recognized regulations on CRC in Current China, and some professional association are actively formulating industry standards and qualification certification for CRC. For example, the Professional Committee of Pharmaceutical Clinical Trials of Guangdong put forward the Guangdong consensus on CRC management, and The Alliance of Pharmaceutical Clinical Trial Institutions also issued CRC industry guidelines. Despite the efforts of the sponsors, hospitals and even SMO itself, there are no clear operational standard for the common acceptance.
The good news is that 7.22 has opened up a new situation of clinical trial quality requirements, but that the road is a long way to go, pursuing of the drug clinical trial is gradual and endless. Only corporation and continuous efforts together can make progress.
The all authors declare that there is no conflict of interest.
- Good Clinical Practice of Pharmaceutical Products, China State Food and Drug Administration (2013). [Crossref]
- Chieko K, Hideo K, Shunsuke O, Naoko K, Kazuyuki S, et al. (2014) High Rate of Awarding Compensation for Claims of Injuries Related to Clinical Trials by Pharmaceutical Companies in Japan: A Questionnaire Survey. 9:e84998. [Crossref]
- Cong YL. (2010). "Comparative Study on the Protection of Chinese and American Subjects in Clinical Trial - from the Perspective of Institutional Review Board." Journal of Peking University (Health Sciences) 42(6): 625-628. [Crossref]






