SARS-COV2 has been shown to be highly thrombogenic owing to the surge of pro-inflammatory cytokines during the cytokine storm requiring therefore high doses of anticoagulant agents. Nonetheless, hemorrhagic events among COVID-19 patients have been well-documented making the prognosis even poorer.
Herein, we describe a case of life-threatening iliopsoas hematoma (IPH) during the clinical course of Covid-19. The patient had been recovering gradually from an acute respiratory failure when he suddenly developed iliopsoas hematoma. The aim of this report is to highlight the severity of this complication as well as the judicious use of anticoagulation therapy during Covid-19.
SARS-COV2; Iliopsoas Hematoma; Prognosis
Thromboembolic prophylaxis is part of the standard management of patients hospitalized for SARS-COV2 pneumonia. In fact, despite adequate thromboprophylaxis, roughly one third of patients critically affected by COVID-19 developed pulmonary embolism [1]. This relatively high rate of thromboembolic complications led to higher doses of anticoagulants depending on clinical complexity and D-dimer levels. However, hemorrhagic complications may arise during the course of the disease such as ilio-psoas hematoma, a rare but serious and potentially fatal complication in the absence of immediate management.
A 67-year-old man, followed for chronic obstructive pulmonary disease (COPD) group B for 7 years, operated for peritonitis by gastric ulcer perforation 26 years ago, chronic smoker and alcoholic weaned 6 years ago; obese with a body mass index of 30Kg/m2; was admitted on July 16,2021 to the medical intensive care unit (P27) of the Ibn Rochd University Hospital in Casablanca for COPD exacerbation due to SARS-COV2 pneumonia going back to 10 days before his admission by the onset of a flu syndrome made of asthenia, myalgia, associated with a dry cough and a diarrhea, the whole evolving in a context of fever which was not correctly measured. The condition worsened 5 days later with the development of dyspnea at rest. A Covid-19 RT-PCR was performed and came back positive, as well as a thoracic CT scan showing a parenchymal damage of about 40-50%. The initial somatic examination noted a conscious patient, 15/15 in the Glasgow Coma scale with symmetrical reactive pupils and without sensitivomotor deficit; polypneic at 33 cpm , with a peripheral capillary oxygen saturation around 65% in the open air rising to 95% under high concentration mask 15L/min; hemodynamically stable with a heart rate of 83 bpm and a systolic blood pressure of 132 mmHg, diastolic blood pressure of 71 mmHg, and mean blood pressure of about 91 mmHg; apyretic at 36.7°c with a capillary blood glucose of 1.57g/l. Pleuropulmonary auscultation showed bilateral rhonchi, cardiovascular auscultation was normal. Abdominal physical assessment showed diffuse abdominal meteorism. The calves were free without oedema of the lower limbs. An ultrasound echocardiogram was performed and showed a global hypokinesia with an estimated ejection fraction of around 45%, a dilated right ventricle with an estimated systolic pulmonary artery pressure of about 55 mmHg, a moderately dilated inferior vena cava of 24 mm. The abdominal ultrasound showed no abnormalities.
The initial biological work-up revealed a natraemia of 133 meq/l, kalaemia: 5.3 meq/l, albumin: 34 g/l, urea: 0.31 g/l, creatinine: 6.9 mg/l, Asat/Alat: 122/99 IU/l, C-reactive protein: 95 mg/l, Procalcitonin: 0.39 g/l, Troponins: 29, 6 UI/l, glycated haemoglobin (HbA1c) : 6, 4%, haemoglobin:15, 2 g/dl , White blood cells: 19150/mm3, Neutrophils: 17980/mm3, lymphocytes: 340/mm3, Platelets: 615000/mm3, Prothrombin ratio: 88%, Activated Cephalin time: 32'', Fibrinogen: 3, 46 g/l, D-dimer: 12350 ng/ml, Anti Xa activity: 1, 21. Therapeutically, the patient was put on a high concentration mask 15L/min with 2 sessions/day of non-invasive ventilation (NIV); a dual antibiotherapy combining Ceftriaxone and Moxifloxacin was also started along with a systemic corticosteroid therapy based on Methylprednisolone, a curative low-molecular weight heparinotherapy at a dose of 10500 units/day of Bemiparin given with Aspirin; a diuretic treatment based on Spironolactone with bolus administrations of Furosemide; Omeprazole double dose; Ivabradine; an alternated nebulizer treatment of Salbutamol/Ipratropium bromide associated with a morning inhalation of Indacaterol/Glycopyrronium bromide; vitamin therapy C, D and Zinc; and respiratory physiotherapy. The clinical evolution was marked, on the 7th day of hospitalization, by a sudden onset of acute abdominal pain with the appearance of diffuse ecchymoses on the right flank as well as the ipsilateral lumbar region. The haemogram showed an acute drop in haemoglobin level from 14.9 g/l to 7.2 g/l. A thoracic-abdominal-pelvic CT scan showed an abundant left hydropneumothorax (figure 1) with an extension of the parenchymal interstitiel damage reaching 75% as well as a pericardial effusion of small abundance; all associated with a thickened aspect of the iliac head of the right Psoas muscle, with an oval formation, fairly well limited, spontaneously hyperdense (50 Hounsfield units), measuring approximately 60x32mm and extending over 108mm, correspondind to a Psoas hematoma (figure 2). Immediate management consisted of ceasing Bemiparin/Aspirin, administration of tranexamic acid as a antihemorrhagic agent, along with blood transfusion and fluid therapy. The evolution was complicated by a hemodynamic instability with an acute respiratory failure for which the patient was intubated and ventilated. Death occurred at day 9 of hospitalization.
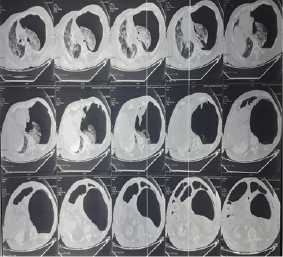
Figure 1. Thoracic Computed Tomography Scan Showing the Left Hydropneumothorax
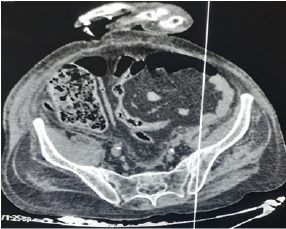
Figure 2. Abdominopelvic Computed Tomography Scan Showing the Hematoma of the Right Iliopsoas Muscle
The overall proportion of patients with bleeding complications during SARS-COV2 infection range from 4.8% to 8%, of which approximately 3.5% had major bleeding [2, 3]. However, critically ill patients with coronavirus disease (COVID-19) are also at increased risk of developing a hypercoagulable state, as clinically evidenced by strokes and high D-dimer levels observed in young patients warranting thus an optimal anticoagulation approach [4]. The pathophysiology of this hypercoagulable condition can be explained by the significant rise of serum pro-inflammatory cytokines during the Covid cytokine storm such as interleukin-6 (IL-6), leukocytic pyrogen or interleukin-1 beta (IL-1β) and tumour necrosis factor alpha (TNF-α), leading therefore to atherosclerotic alterations [5]. These pro-inflammatory cytokines have also been shown to be one of the causes of disseminated intravascular coagulation (DIC) observed in septic patients as well [6, 7]. Therefore, to combat this hypercoagulable state, patients with COVID-19 should be properly anticoagulated to minimize thrombotic and ischemic risk. Nevertheless, anticoagulant therapy coupled with an antiaggregant agent such as aspirin increases markedly the risk of major bleeding complications such as retroperitoneal hemorrhage [6]. High-dose heparin during Covid-19 may not only be ineffective, but might also be harmful by potentially contributing to the bleeding component of the cytokine microangiopathy [8]. In fact, the pathophysiology of psoas hematoma in critically ill patients with covid-19 remains not fully elucidated. Severe coughing, a common symptom of covid-19, may cause a significant rise in intra-abdominal pressure leading to bleeding by arteriolar rupture [9]. The increase in intra-abdominal pressure due to non-invasive ventilation and potentially worsened by coughing could also lead to arterial rupture and consequently retroperitoneal bleeding. Similarly, the use of high positive expiratory pressures during invasive ventilation for patients enduring a severe acute respiratory distress syndrome may, through intra-abdominal pressure, leads to retroperitoneal bleeding by arterial rupture. Abdominopelvic computed tomography scan remains the initial imaging of choice in case of suspected retroperitoneal hemorrhage. Immediate management consists of stopping anticoagulant agents, transfusion of compatible red blood cells, optimal vascular filling, along with adjuvant support measures [10]. Hemodynamically unstable patients can be treated by arterial embolization as it is minimally invasive with rapid therapeutic efficacy compared to surgical treatment. In case of active bleeding, arterial embolization of the third and fourth lumbar arteries is usually warranted while taking into account its technical challenges as it requires catheterization with selective embolization of the bleeding artery, as well as post-embolization evaluation to document the success of treatment.
Anticoagulant therapy, although indicated in COVID-19 patients, has its own drawbacks with few guidelines directing indications and dosages in this disease. Randomized clinical trials would therefore be of paramount to evaluate the benefit-risk balance of curative versus prophylactic heparin therapy during Covid-19 course. To sum up, it should be emphasized that any acute drop in hemoglobin level during the evolution of covid-19 with an eventual hemodynamic instability should suggest a psoas hematoma warranting thus a diagnostic abdominopelvic CT scan. Discontinuation of anticoagulation, administration of hemostatic agents, blood transfusion, and optimal fluid therapy are the systematic initial measures. In case of persistent bleeding, hemostasis would preferably be performed by selective embolization or, if necessary, by conventional surgery.
- Zerbato V., Bozzato AM., Di Bella S., Giuffrè M., Martingano P., et al. (2021) Spontaneous psoas haematoma: a life-threatening complication of anticoagulation in COVID-19. A case series of four episodes. Infect Dis (Lond) 53: 724-729. [Crossref]
- Al-Samkari H (2020) COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood 136: 489–500. [Crossref]
- Shah A (2020) Thrombotic and haemorrhagic complications in critically ill patients with COVID-19: a multicentre observational study. Crit Care 24: 561. [Crossref]
- Cattaneo M., Bertinato EM., Birocchi S., Brizio C., Malavolta D., et al. (2020) Pulmonary embolism or pulmonary thrombosis in COVID-19? Is the recommendation to use high-dose heparin for thromboprophylaxis justified? Thromb Haemost. [Crossref]
- Zhou F., Yu T., Du R., Fan G., Liu Y., et al. (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395: 1054–1062. [Crossref]
- Marty B., Sanchez LA., Wain RA., Ohki T., Marin ML., et al. (1998) Endovascular treatment of a ruptured lumbar artery aneurysm: case report and review of the literature. Ann Vasc Surg 12: 379-383. [Crossref]
- Wada H., Tanigawa M., Wakita., Nakase T., Minamikawa K., et al. (1993) Increased plasma level of interleukin-6 in disseminated intravascular coagulation. Blood Coagul Fibrinolysis 4: 583-590. [Crossref]
- Bikdeli B., Madhavan MV., Jimenez D., Chuich T., Dreyfus I., et al. (2020) COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC State-of-the-Art review. J Am Coll Cardiol 75: 2950-2973. [Crossref]
- Amole AO., Kathuria MK., Ozkan OS., Gill AS., Ozkan EO (2008) Lumbar artery laceration with retroperitoneal hematoma after placement of a G-2 inferior vena cava filter. Cardiovasc Intervent Radiol 31: 1257-1259. [Crossref]
- Hamid RS., ul HT., Chishti I., Azeemuddin M (2010) Post traumatic avulsion of lumbar artery: a rare cause of retroperitoneal haemorrhage treated by glue embolization. J Pak Med Assoc 60: 487-489. [Crossref]


